Women's Health Screening _Quo Vadis?
The recent Federal Government decision to rebate cervical cancer screening only every 5 years and to only rebate for screening after the age of 24 years and under the age of 74 years raises the question as to whether other aspects of women's health could be also screened far less frequently. That could be an unintended negative consequence of the recent Government funding decision.
The chance of an Australian woman being diagnosed with a gynaecological cancer by age 85 is 1:5. At its best a well women's check not only screens for cervical cancer (1% incidence) but also for:
- Breast cancer (1:8 or 12%)
- Uterine cancer (2.5%)
- Ovarian cancer (1.8%)
- Vulval cancer (0.1%)
- Benign ovarian tumours and cysts
- Lichen sclerosis (0.3%) of whom 4% develop vulval cancer
- Uterine fibroids (40% >aet 40)
- Utero-vaginal prolapse (25%)
- Urinary incontinence (37%)
- Anal incontinence (12.9%)
- Severe menopausal symptoms (22%)
- Osteoporosis (20%> aet 50)
The value of early diagnosis is in improving survival rates. The survival from most gynaecological cancers is stage dependent:
- Cervical Cancer: Progresses at a rate of - 1 Grade per annum. The 5 yr survival rate by the stage of the cancer approaches 93% for stage 1A, 80% for Stage 1B, 63% for stage 2A, 35% for stage 3A, 16% for stage 4A.
- Uterine cancer: The 5 yr survival rate by the stage of the cancer approaches 85% for stage I, 75% for stage II, 45% for stage III and 25% for stage IV.
- Ovarian Cancer: The 5 yr survival rate by the stage of the epithelial cancer approaches 90% for stage 1; 70% for stage 2; 39% for stage 3; 17% for stage 4. Unfortunately most ovarian cancers are diagnosed at a late stage. A current view is that many ovarian cancers arise from Fallopian tubes - hence the new emphasis on salpingectomy at hysterectomy.
- Vulval Cancer. The survival rate by the stage of the squamous cancer approaches 93% for stage 1; 79% for stage 2; 53% for stage 3 and 29% for stage 4
- Vaginal Cancer: The survival rate by the stage of the squamous cancer approaches 84% for stage 1; 75% for stage 2; 57% for stage 3&4.
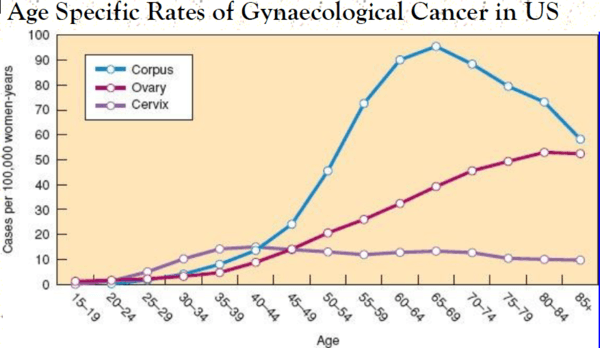
The above graph demonstrates the importance of screening for uterine corpus cancer and ovarian cancer in the peri-menopausal and postmenopausal age groups. The tools available are imperfect however :
- Uterine corpus cancers can be predicted using ultrasonic endometrial thickness >5 mm in the postmenopausal age group (especially in menopausal diabetics and Tamoxifen treated patients who are at greater risk)
- Ovarian tumour markers have been improved using a risk of ovarian malignancy algorithm (ROMA) based on the levels of CA125 and HE4. Furthermore BRCA gene testing may stratify risk in those with a family history.
Savings Expected by New Cervical Screening Program (CSP)
After implementation of the National Cervical Screening Program in 1991, incidence of cervical cancer declined by 36%, and mortality by 44%, by the mid-2000s. In Australia, the emergent evidence on HPV screening in conjunction with the introduction of HPV vaccination, and the comparatively longer screening intervals and narrower age range for screening recommended by the International Agency for Research on Cancer prompted a major review of the National Cervical Screening Program. The annual cost of the National Cervical Screening Program was estimated to be AUS$215 million in 2015. The estimated saving of the new program using HPV screening is 26%. What is uncertain is the national cost of delays in the diagnosis of non-cervical gynaecological cancers as a result of the reduced incentive to visit the doctor more frequently than every 5 years because of the new CSP.
Further reading:
1.Ferley J, Shin HR, Bray F, et al. GLOBOCAN 2008 v1.2, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 10 International Agency for Research on Cancer; [October 5, 2011]. 2010. Available from: http://globocan.iarc.fr.
2. Lew J-B et al Primary HPV testing versus cytology-based cervical screening in women in Australia vaccinated for HPV and unvaccinated: effectiveness and economic assessment for the National Cervical Screening Program The Lancet 2017 2(2):e96-e107
| Tags:Other |








